AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
 Other types of crystalline SEs including oxide-based SEs (e.g., perovskite-type 9, sodium superionic conductor (NASICON)-type 10, and garnet-type 11, 12) and halide-based SEs (e.g., Li-M-Cl system, M = Y, In, Sc 13, 14, 15, 16, 17) also demonstrate good conductivities of 10 −4–10 −3 S cm −1. For example, representative sulfide-based SEs, such as Li Argyodites 5, 6 and Li 10GeP 2S 12 (LGPS)-type 7, 8, exhibit attractive ionic conductivities in the order of 10 −2 S cm −1. 
Crystalline SEs with long-range ordered structures have shown continuous and fast Li-ion conduction. One of the essential requirements for a favorable SE is high ionic conductivity. A key component for ASSBs is solid electrolyte (SE) which can potentially enable the use of high-voltage cathodes and Li metal anode to boost the energy density 3, 4. Long cycle life (more than 2400 times of charging and discharging) can be achieved for all-solid-state batteries using the xLi 2O-TaCl 5 amorphous solid electrolyte at 400 mA g −1, demonstrating vast application prospects of the oxychloride amorphous solid electrolytes.Īlong with the fast growing market of rechargeable electric vehicles (REVs), the development of all-solid-state batteries (ASSBs) is of high expectation due to their promises of safety, reliability, and high energy density 1, 2. More importantly, all-solid-state batteries using the amorphous solid electrolytes show excellent electrochemical performance at both 25 ☌ and −10 ☌. It is found that the oxygen-jointed anion networks with abundant terminal chlorines in xLi 2O-MCl y amorphous solid electrolytes play an important role for the fast Li-ion conduction. The mixed-anion structural models of xLi 2O-MCl y amorphous SEs are well established and correlated to the ionic conductivities. xLi 2O-MCl y amorphous solid electrolytes can achieve desirable ionic conductivities up to 6.6 × 10 −3 S cm −1 at 25 ☌, which is one of the highest values among all the reported amorphous solid electrolytes and comparable to those of the popular crystalline ones. Herein, we report a new family of amorphous solid electrolytes, xLi 2O-MCl y (M = Ta or Hf, 0.8 ≤ x ≤ 2, y = 5 or 4). Positions are measured using the meter sticks, then wavelengths are determined from the positions using the graph itself or the equation of the best fit line for that graph.įor atoms that contain only one electron, the theory of atomic structure proposed by Niels Bohr can be used to calculate wavelengths for transitions between particular electronic energy levels of the atom.Solid electrolyte is vital to ensure all-solid-state batteries with improved safety, long cyclability, and feasibility at different temperatures. The calibration graph is therefore an integral part of the spectroscope. For example, using the same apparatus and without moving the relative positions of the meter sticks, diffraction grating and lamp, it is possible to view the spectrum of a new element, measure where its spectral lines occur on the meter stick, and then read the graph or use the equation of the line to determine the wavelength to which each of those positions corresponds. Once the best fit straight line has been determined, the equation of this line can then be used to convert positions of other spectral lines to wavelength. 
position of the spectral line will yield a straight line. Since this position depends upon the wavelength in a linear way, a graph of wavelength vs. Using a light source that contains known wavelengths of light, we can measure exactly where each known wavelength appears along a meter stick. As the light emerges after being reflected by the grating, these tiny lines cause the reflected light to interfere with itself in such a way that the different wavelengths of the light to appear in different positions to the left and right of the original direction in which the light was traveling. 
A diffraction grating is a piece of glass or clear plastic with many very narrow and closely spaced lines on it. If we view the light through a prism or a diffraction grating, however, the individual wavelengths are separated. 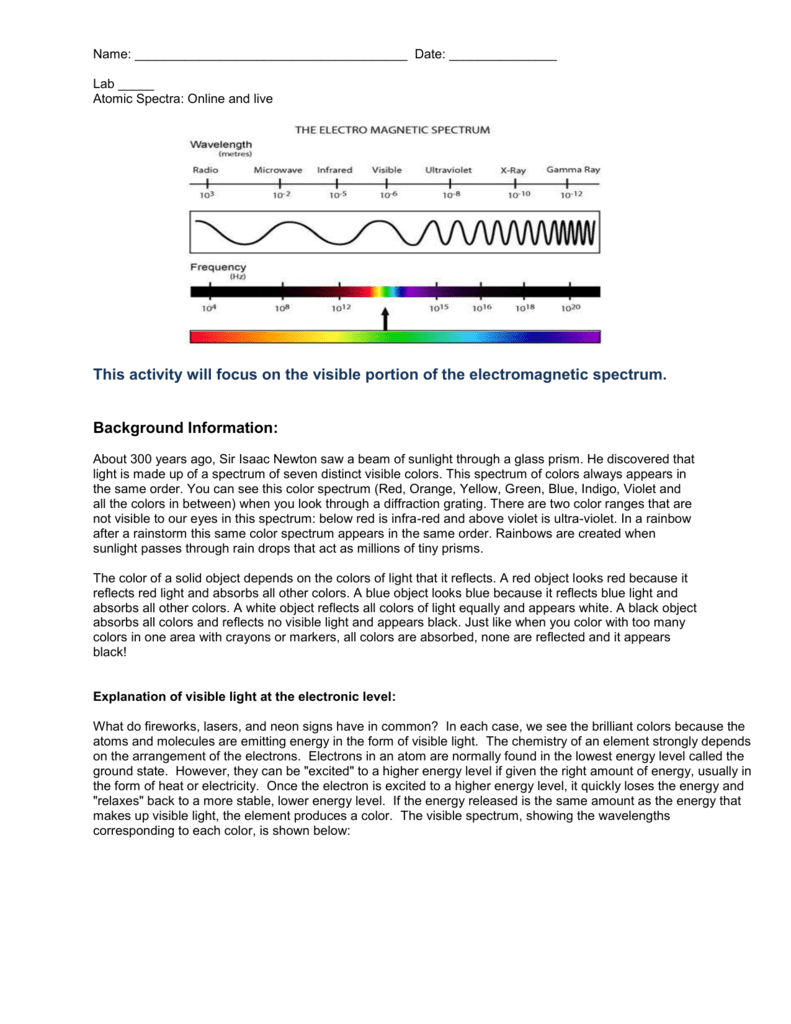
To the naked eye, the various wavelengths (colors) of light emitted by an element are mixed together and appear as a single color that is a combination of the component colors. To measure these wavelengths in the laboratory, we must first separate them. Thus, the spectrum of an element can be stated by listing the particular wavelengths of light that its atoms emit. These two relationships combine to give a third: 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
